To monitor inter-subunit rotation during ATP hydrolysis, we attach two fluorophores - in the first approach Cy5 (red) at the rotating g-subunit (blue helices) and tetramethylrhodamine (green) at one of the two b-subunits - and reconstituted the enzymes fully functional into liposomes [1].
Fluorescence resonance energy transfer (FRET) is monitored in photon bursts of freely diffusing ATP synthases with a confocal setup for single-molecule detection.
Incubation with AMPPNP results in stable intensity ratios within a burst and three different FRET efficiencies, which correspond to three possible g-subunit orientations with respect to the b-subunits.
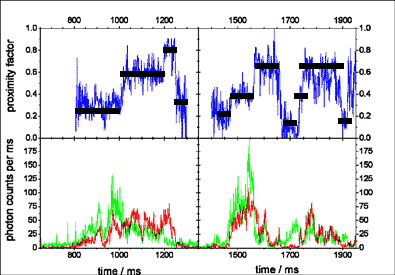
During ATP hydrolysis, a consecutive order of three distinguishable FRET efficiencies (steps in the proximity
factor traces, blue) was observed
indicating a stepwise movement of the g-subunit. The dwell times of the FRET levels observed in this FRET approach are correlated to the catalytic rates.
The sequence of FRET level transitions was reversed as the conditions were changed from ATP hydrolysis to ATP synthesis. This was the confirmation of the opposite direction of rotation (Nature Structural & Molecular Biology 02/2004).
The FRET approach was refined and demonstrated an conformational change of the e-subunit associated with the catalytic activity of the enzyme (EMBO Journal 06/2005).
Subsequently we localized the C terminus of the proton channel (subunit a) in the Fo part by FRET triangulation. A single rotating FoF1 showing three 120° steps in about 100 ms provided the basis for three dimensional reconstruction of the asymmetric position of subunit a (J Biol Chem 11/2008).
We are using a FRET approach to observe the rotation of a single membrane-embedded Fo-motor during catalysis
(EMBO Journal 09/2009), and we are developing a nano-electrochemical tool to control the rotation and conformational state of individual subunits in
a single ATP synthase.
As the lastest step we have now implemented a triple-FRET
detection scheme using three fluorophores and a sequence of three alternating lasers to simultaneously monitor the F1 and the Fo motor in a single ATP synthase.
Ultimately we use new FRET labeling tools for rotation measurements of a single ATP synthase in a living E. coli cell. As the driving forces for ATP synthesis differ from our previous in vitro measurements, we want to answer the question how a few hundred enzymes manage to maintain the cellular [ATP] level in bacteria.